By Rachel Leea,b James E Simona,c, William Reichertc, H. Rodolfo Juliania,c and Beverly J Teppera,b
Sweet basil is an aromatic herb with myriad of uses as an ornamental plant, culinary flavoring and bioactive aroma ingredient in foods, cosmetics and household products. The demand for fresh basil continues to grow, especially for new varieties with unique aroma and chemical characteristics. Nevertheless, what is desirable in fresh basil for cooking may not be suitable for a hand cream or scented candle, which may require a different aroma/chemical profile. Linking the volatile chemical composition of basil to the human sensory experience is a necessary first step in guiding the efforts of plant breeders, plant growers, ingredient suppliers and product developers in fulfilling the diverse needs of the marketplace for fresh basil and basil-scented products. Using a trained descriptive panel, we developed a lexicon (standardized sensory vocabulary) for fresh basil aroma and linked the aroma profiles with the main volatile components quantified by GC-MS headspace analysis. Here we report initial results for five basil varieties including Queenette Thai, Sweet Dani (lemon basil) and three Rutgers experimental breeding lines MC-9 (cinnamon), Italian basil-1 (SB-22) and Italian Basil 2 (CB-39). According to the trained panel, Queenette Thai basil was strongly anise-like (high in methyl chavicol), Sweet Dani was strongly lemon-like (high in citrus volatiles) and the cinnamon basil was rated high in methyl cinnamate aroma (matching its high methyl cinnamate content). The two Italian basils differenced in their aroma and chemical profiles. Italian Basil 2 was predominantly anise-like (methyl chavicol) with the presence of other spice notes (eucalyptus/clove). Italian Basil 1 had a more complex profile described as spicy (generic spice) with aromas of eucalyptus/clove and some anise. Both Italian basils had high concentrations of sweet/floral compounds (e.g., linalool) consistent with their identities as ‘sweet’ cultivars.Future studies will expand the lexicon to include taste, texture and flavor-by-mouth attributes to obtain a comprehensive sensory description of fresh basil.
Characteristics and Uses of Sweet Basil
Sweet basil (Ocimum basilicum) often sold under the commercial names “Italian” basil, “Genovese” basil or “Large-leaf” basil and others is the most economically significant culinary herb in the U.S. and Europe [9;45]. Basil originates from Southern Asia and Africa [32] and is a member of the Lamiaceae family, which includes lavender, mint, rosemary, sage and thyme. This highly aromatic plant has numerous culinary uses [41-43]. Fresh and dried basil leaves are prominent flavoring ingredients in North American, Mediterranean and Southeast Asian cuisines. Basil essential oils and dried leaves are also used in the food industry to flavor liqueurs, confections, meat products, baked goods and tomato-based products. The flowers are also used to relish cheese, butter, oil, vegetables and fish [34]. Thai basil seed is also used in popular desserts and soft drinks consumed in the Middle East and Asia.
Basil also has a long history of use in traditional medicines and religious ceremonies due to a rich abundance of bioactive compounds [43] including antibacterial, antifungal and insecticidal activities [27;30]. Basil may also lower the risk of liver disease and ulcers and has been used to treat gastrointestinal disorders [16;27] and exhibits antiviral [5] and other pharmaceutical properties [28]. Due to high concentrations of flavonoid and phenolic compounds including rosmarinic acid, basil exhibits strong antioxidant activity [11;19;22]. Anthocyanin-rich basils exhibit other health related activities [33]. New commercial uses for basil continue to be explored; increasingly, basil extracts and essential oils appear in cosmetics, skin creams and lotions [40].
Major Chemical Volatiles
Basil essential oils are complex mixtures of components, with more than 200 chemical compounds having been identified [2;3;20]. The plant accumulates monoterpenes, sesquiterpenes and phenylpropenes in the glandular trichomes located on the surface of the leaves and flowers [7;10;12]. The main components are oxygenated monoterpenes and phenylpropane derivatives [15]. Yet, there is significant variation in the constituents in the essential oils. Major monoterpenes of sweet basil include (R)-linalool and 1,8-cineole that characterize its sweet and eucalyptus-like sensory qualities, respectively [43]. The main phenylpropenes, eugenol and methyl chavicol [10] characterize its clove-like and anise-like aromas [43].
Other major chemical constituents in the essential oils of basil contribute to its bouquet, and the presence and relative ratio of these major and minor volatile constituents such as linalool, methyl chavicol, citral, eugenol, methyl cinnamate and others impart unique aromas and flavors [4;8;41]. Sesquiterpenes are usually found in minor amounts, the main ones include germacrene D, (E) caryophyllene, α-guaiene, bergamotene and α-humulene, among others.
In purple basils, α-guaiene can reach levels up to 7% (20). Italian large leaf basil contained minor amounts of α-humulene (0.4%), germacrene D (2.1%), bicyclogermacrene (0.8%) and d-cadinene (1.2%), whereas, sweet basil contained high levels of germacrene D (5.3%), bicyclogermacrene (2.2%), α-guaiene (2.4%) and α-amorphene (3.26%), with total sesquiterpenes up to 14%. Sweet Dani lemon basil showed a different profile and higher levels of sesquiterpenes (up to 16%) characterized by high levels of (E) caryophyllene (5%), germacrene D (7%), α- and β-selinene (2% each) (20).
Age and Location of Leaf on the Plant Affects Essential Oil Profile
The concentration of essential oils may also differ among leaves depending on the age of the leaf and location on the stem. A study by Fischer et al. [10] evaluated variation in essential oil composition of the sweet basil cultivar “Perrie” within individual leaves by evaluating leaf position and leaf age. The goal of the study was to understand the dynamics of essential oil composition in individual leaves, as influenced by leaf development and age, by recording kinetics in individual leaves of the plant. At 16-weeks-old, this cultivar has a main stem with up to 11 nodes and leaf age is dependent on growth position, both laterally and vertically.
Results showed that on the main stem, higher essential oil concentrations were found in younger leaves for 33 essential oil compounds, including monoterpenes linalool and 1,8-cineole [10]. Also, leaves positioned closer to the plant tip had higher essential oil concentrations than older leaves and leaves closer to the root. On the lateral shoots, the percentage of eugenol in essential oil extracts was higher than the percent of methyleugenol in all leaves, including leaves growing on the main stems. Methyleugenol concentrations were also higher from older leaves closer to the stem than from younger leaves farther from the stem. Linalool was lower in mature leaves than in younger leaves. These findings suggested that eugenol was converted to methyleugenol and linalool decreased as the leaf matured [10]. Therefore, even within the same basil variety, changes in the aroma profile will occur due to leaf age, growth rate and because of the nature of terpene and phenylpropene bioaccumulation over time [7;8;12]. It is important to evaluate basil leaves at similar maturity, growth rates and similar environmental conditions across a study because each of these factors can impact essential oil composition and therefore the aroma.
Classification of Basil Varieties Based on Chemotypes
As many as seven chemotypes have been identified within the Ocimum basilicum spp. that may be important in differentiating basil varieties and their sensory profiles [13;25;46]. In a recent study [46], 38 genotypes of sweet basil were evaluated and grouped into seven chemotypes characterized by high concentrations of: 1) linalool; 2) linalool-eugenol; 3) methyl chavicol; 4) methyl chavicol-linalool; 5) methyl eugenol- linalool; 6) methyl cinnamate-linalool; and 7) bergamotene, a sesquiterpene, characterized by a woody aroma.
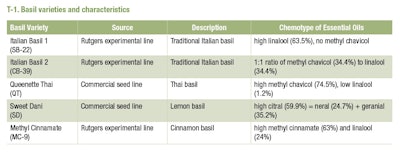
Varieties of basil are bred for a wide range of purposes including cold tolerance, disease resistance, and/or different bouquets of aromas [35-37;45]. These characteristics are reflected in their specific chemotypes. For example, ‘Queenette Thai’ is a commercial variety with a high concentration methyl chavicol (group 3 chemotype) that has a strong anise-like aroma but little sweetness. In contrast, ‘Sweet Dani’ is a commercial variety of lemon basil, specifically bred for its high citral content and strong citrus aroma and flavor [31]. ‘Sweet Dani’ is a distinctive variety that adds another unique chemotype to the list of known chemotypes.
Previous Attempts to Relate Basil Chemistry to Human Sensory Perception
There is a wealth of information on the volatile chemical composition of essential oils of basil with limited data linking volatile composition of fresh basil leaves with human sensory perceptions.
In one study [39], essential oils were extracted by steam distillation from six different portions of the plant (leaf, flower, stem, whole plant, fresh leaf-flower, and leaf-flower). Gas Chromatography-Mass Spectroscopy (GC-MS) was used to measure and characterize the volatiles present in the headspace and then linked with the GC sniffing method to describe the human sensory impact. Fifty panelists provided descriptors for 40 compounds identified by GC-MS, yet the aroma descriptors did not capture the major volatiles with precision. The panel described methyl chavicol with a variety of overlapping terms including, ‘fragrant,’ ‘sweet,’ ‘cooling’ and ‘fresh minty’ [39]. However, methyl chavicol is recognized as having a distinctive, anise-like aroma [44]. Further, some odors were described as ‘sour,’ an attribute typically associated with taste rather than aroma perception. Interestingly, the essential oil with the highest concentration of methyl chavicol, (coming from the fresh leaf-flower) was judged the most pleasant, while the oil with lowest concentration of methyl chavicol (from the stem) was the least pleasant [39]. This variability in the chemical profile is consistent with the findings of Fischer et al. [10] showing that various parts of the plant, especially younger leaves have a greater abundance of aroma compounds.
A second study developed a sensory profile of hybrid basil essential oils. However, the list of descriptors (basil, citric, and sweet) was too short to describe the complex aroma profiles of basil and failed to effectively discriminate different cultivars [6].
Finally, Bernhardt et al. [2] used a trained panel to assess sensory differences among 12 varieties of dried basil leaves and flowers and then used GC-MS to quantitate the essential oils from these same samples. A total of 12 attributes were evaluated with four attributes describing appearance (color intensity, color hue, leaf curl and venation) and eight attributes describing aroma (global aroma impact, anise, lemon, hay-like, spicy, sweet, basil and pine resin). The attributes ‘global’ and ‘basil’ are too general to differentiate among closely-related cultivars (chemotypes). Importantly, results showed only five of the cultivars were different from each other and the differences were based primarily based on appearance rather aroma characteristics.
Further, 71 chemical compounds were identified by GC analysis and plotted with the sensory descriptors using Multiple Factor Analysis (MFA). The plot showed close associations among some chemical components of the cultivars and the sensory descriptors (e.g., the presence of high linalool content in a cultivar with high lemon aroma impact). However, major chemical species such as 1,8-cineole and methyl eugenol did not strongly associate with the cultivars on the plot, and methyl chavicol, a signature compound in basil was not associated with anise aroma intensity [2].
Filling Gaps in Knowledge
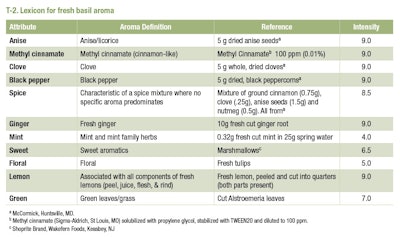
None of the above-mentioned studies, used clearly defined, standardized vocabulary to describe basil aroma, and information was lacking on sensory panel training and performance. Most of the aforementioned published work examined essential oils extracted from fresh and dried basil. One notable exception is the McCormick Spice Wheel that was developed by a highly-trained panel to describe the sensory attributes of dried herbs and spices, including basil [24]. Currently, there is no standard lexicon for fresh basil aroma.
The ability to capture headspace aromatic volatiles with new generation GC/MS Triple Quad systems such as the Shimadzu TQ8040, makes it easy and inexpensive to link natural aromatic volatile emissions to human sensory responses, bypassing the traditional systems of recovering essential oils. These systems include hydrodistillation, steam distillation, solvent extraction, super critical fluid extraction and other approaches that can lead to differential outcomes.
The current study used a trained panel to develop a lexicon for fresh basil aroma that focused on describing the major scent characteristics of different basil chemotypes. The panel then used the lexicon to construct aroma profiles for five fresh basil varieties. Finally, the sensory profiles were linked with the chemical profiles to better understand the impact of the chemical constituents on sensory perceptions.
Methods
Descriptive Analysis
The panel was established and trained using the quantitative descriptive analysis method, first developed by Tragon Corp. in Red Wood Shores, California. Panelists were recruited, trained and monitored for performance using standard procedures outlined in Meilgaard et al. [29]. The panel developed the list of sensory terms (lexicon) and definitions by consensus, and reference standards were established for each sensory term [23]. The panel received approximately 30 hr of training over a six-month period. The panel was monitored for performance using statistical procedures described in the statistical analysis section. Eight panelists out of the original 11 panelists met the performance criteria and participated in the final evaluations. The list of descriptors, their definitions and reference materials are listed in T-1.
Sample Selection and Preparation
Five varieties of Ocimum basilicum were studied based on variations in their essential oil composition. Queenette Thai’ (QT) and ‘Sweet Dani’ (SD) are commercial varieties whereas SB-22 (Italian Basil-1) CB-39 (Italian Basil-2) and MC-9 (cinnamon basil) are Rutgers University Experimental Lines. The major, chemical, constituents of these varietals were previously described by Pyne et al. [36] using GS-MS headspace analysis (T-2).
Four plants of each variety were grown from seed under similar conditions in the New Jersey Agricultural Experiment Station greenhouses, New Brunswick NJ. The plants were grown on a staggered schedule such that two plants of each type were used for training and two plants were used for formal evaluations, which were conducted at approximately 16-weeks maturity.
The plants were transported from the greenhouse to the Food Science Sensory Laboratory 30 min. before each session. Samples for sensory testing were prepared 20 min. before panelist evaluations. Since the concentration of essential oils differs among leaves depending on the age of the leaf and position on the stem [10], young, mature leaves from the top of the plant were selected to minimize leaf-to-leaf variation. Leaves for headspace analysis were harvested at the same time as leaves for sensory analysis.
All samples were separated and identified on a Shimadzu Gas Chromatograph 2010 Plus with a Shimadzu TQ8040 Mass Spectrometer. Headspace sampling was performed on a Shimadzu AOC-6000 auto sampler. Basil dried leaves (50 mg) were placed in headspace vials (20 ml) and were allowed to pre-equilibrate at 70°C for 5 min. in the oven at 250 rpm to ensure reproducibility of the headspace automatic injector, then 100 uL of the headspace were injected into the GC/MS inlet (250°C). Samples were separated on column SH-Rxi-5Sil MS heated from 35°C with a hold of 4 min. to 250°C with a hold of 1.25 min. at 20°C/min., with a total run time of 16 min. Individual, compound, identifications were made by matching spectra with those from a mass spectral library. The identity of each compound was confirmed by its Kovats index [20]. The ion source temperature was set to 200°C, the interface temperature was set to 250°C, the solvent cut time was 3.5 min. and the detector voltage was set to 0.2 kV with a threshold of 1,000.
Panel Procedures and Data Analysis
All five varieties were used at various stages during group training sessions. Formal evaluations were conducted when panelists were seated in individual, sensory booths equipped with RedJade data collection software (Tragon, Corp.). The sessions were conducted under white lights.
Panelists were presented with one whole basil leaf of each variety placed in a two ounce soufflé cup with a four-digit blinding code. They were instructed to rip the leaf in half along its main axis to release the volatiles and to smell the sample ortho-nasally. Panelists waited for 1 min. in between samples. Ratings were collected using 15-cm line scales where 0=weak and 15=strong.
The five basil samples were evaluated in random order within a session and the session was repeated once. Data from the two sessions were averaged and two-factor analysis of variance (ANOVA) was used to assess sample effects, panelist effects and the panelist* sample interaction. In the ANOVAs, sample effects were tested against both error and interaction (panelist* sample). The latter procedure adjusts for variability in sample ratings across panelists and the more conservative outcome from the two tests was used. ANOVA calculations were followed by Duncan’s Multiple Range Test to probe for differences among sample means. Panel performance was assessed using RedJade diagnostic tools, and by examining panelist effects, replicate effects, and panelist* replicate interactions using SAS statistical analysis software (Version 9.4, The SAS Institute, Cary, North Carolina).
Analysis and integration of the GC/MS samples was performed using GCMS solution v4.3a. Individual compound ID’s were determined by comparing our results with current literature and screening them against the NIST05.LIB, NIST05s.LIB, W10N14.lib and the W10N14R.lib mass spectral libraries with a > 90% similarity search.
Results and Discussion
Sensory Aroma Profiles
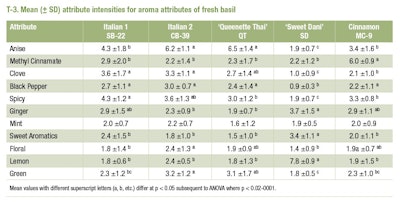
The five basil varieties exhibited different sensory profiles, varying in all attributes except for mint aroma, which did not differ across varieties. The results show that Queenette Thai basil was predominantly ‘anise-like’ with spice, clove and green notes, while Sweet Dani was primarily ‘lemon-like’ with moderate ginger and sweet aromatics. Aromas of spice and black pepper were noticeably absent in this variety. The cinnamon basil (MC-9) was primarily ‘cinnamon-like’ with other spice notes present. The two Italian basils showed many similarities in their aroma profiles. Both varieties had notes of clove, black pepper and general spice. However, Italian Basil 2 had a pronounced ‘anise-like’ aroma and more green and floral aromas than Italian Basil 1. Differences in the mean values for all attributes are also shown in T-3.
Panel Performance
Metrics for panel performance were high. Panel mean standard deviations across attributes ranged from 0.8-1.5 and 0% of panelists exceeded the panelist standard deviation score > 2.5. We observed panelist effects for all attributes and most attributes showed panelist* sample interactions (p-value range = 0.04-0.001). Training reduces variation among panelists, yet differences among panelists in sensitivity, response behavior to stimuli and other unknown factors remain regardless of the training method or level [26]. Inherent differences in aroma perception reflect human genetic variability [17;21] and variability across plants, even when they are grown under controlled greenhouse conditions. Variability in horticultural products is a well-known source of error in panel assessment [1] making differences between samples more difficult to detect [14] .
The ability of panelists to score the attributes consistently over sessions is captured in the replicate effects [38]. Non-significant replicate effects indicate high rates of consistency. For the most part, replicate effects were not statistically significant except for the attribute methyl cinnamate, which just reached the statistical cutoff (p<0.044).
Chemical Composition
Chemical profiles of the five varieties differed based on head-space analysis. The Appendixshows the complete chemical profile for each basil variety studied including retention time, relative percentages, and aroma descriptor for each chemical component. The aroma descriptors were previously identified in the literature [31;41;44].
F-2 shows the major chemical constituents for each basil sample. Queenette Thai had a high percentage of methyl chavicol (69%), accompanied by the presence of 1,8-cineole (17%); linalool was absent from this sample.
Sweet Dani basil had high citral and citral intermediates (65%) and lacked linalool and 1,8-cineole. Cinnamon basil contained methyl cinnamate (26%) and linalool (41%), with 1,8-cineole as a minor constituent (3.8%). Italian Basil 2 had comparable amounts of methyl chavicol (29%), linalool (22%) and 1,8-cineole (19%). Italian Basil 1 had no methyl chavicol, but did have a high concentration of linalool (47%) and 1,8-cineole (25%). These chemical profiles are consistent with the known chemotypes of these basils as described in T-2.
It is important to note eugenol, the primary constituent of ‘clove-like’ aroma, was mainly absent in the varieties studied. However, 1,8-cineole, a fresh, eucalyptus-like aroma was present in all the samples except Sweet Dani. The rank-order of 1,8-cineole concentration in the samples was: Italian Basil 1 (25.0%) > Italian Basil 2 (19.0%) > Queenette Thai (14.0%) > Cinnamon Basil (3.8%) > Sweet Dani (0%). This was precisely the same order the panel rated the samples in clove intensity. Thus, it seems reasonable to assume the panel rated this attribute accurately, albeit with the wrong label. Since many plant species contain both compounds, it is not surprising this attribute could cause sensory overlap for the panelists.
Comparison Between the Sensory and Chemical Profiles
To better visualize the results, the chemical composition data for each variety was organized into a pie chart and described using the sensory attributes (F-3, F-4) For ease of interpretation, some of the attributes were combined, e.g., floral and sweet, fresh/eucalyptus and clove.
Overall, the panel profiles were in line with the chemical profiles of the samples. Queenette Thai contained an abundance of methyl chavicol and was rated high in anise aroma by the panel. Sweet Dani was high in citral and related compounds and was rated predominantly lemon-like by the panel. The cinnamon basil (MC-9) contained a high concentration of methyl cinnamate and was rated high in methyl cinnamate aroma (see F-3). The cinnamon basil also had a high concentration of linalool (sweet/floral aroma), but this characteristic was not obvious in this sample. Interestingly, an untrained panel in a previous study rated the aroma of methyl cinnamate to be much less pleasant than that of cinnamaldehyde [18]. Methyl cinnamate is typically described as cinnamon-like, but it lacks the sweetness of cinnamaldehyde, which is the primary volatile in sweet, cinnamon spice [41]. It is possible that the combination of high methyl cinnamate in this sample along with the presence of woody and green notes obscured the perception of its sweetness.
The panel also discriminated the two Italian basils in accordance with the chemical profiles of these varieties (see F-4). Italian Basil 2 (CB-39) was predominantly anise-like (methyl chavicol) with the presence of other spice notes (eucalyptus/clove). In contrast, Italian Basil 1 lacked the singular intensity of anise aroma of Italian Basil 2. However, this sample had a more complex profile described as spicy (generic spice) with aromas of eucalyptus/clove and some anise. Both Italian basils had high concentrations of sweet/floral compounds (e.g., linalool) consistent with their identities as ‘sweet’ cultivars. It appears the panel incorporated these sweet/floral notes into their ratings of the spice aromas rather than rating them as a noticeable, separate percept.
Conclusions
The present data provide an important starting point for establishing standardized vocabulary for the description of fresh basil aroma. Future work will extend the lexicon to include the full complement of appearance, taste, texture and flavor-by-mouth attributes. Future efforts will also examine a greater number and diversity of varieties. The current study was limited to five varieties, which were either lacking in or not perceptually different in attributes such as woody, mint and earthy, known characteristics of other sweet basil cultivars. Although chemical compounds imparting some of these qualities (e.g., woody) were present as minor constituents in the samples, their levels might not have been high enough to reach perceptual thresholds. A concise lexicon will inform the design of consumer studies to assess consumer preferences for fresh basil for culinary uses and will assist the industry in selecting varieties with specific traits for a range of consumer products.
Acknowledgments:
This research was supported by funds from the Office of Research and Economic Development at Rutgers University, the Rutgers Center for Sensory Sciences and Innovation (CSSI), Symrise Inc., Teterboro, NJ, and the New Jersey Agricultural Experiment Station and HATCH project reports NJAES 12131 and 1005685. Aroma volatiles from basil were captured and analyzed using a Shimadzu Instruments TQ8040 GC/MS Triple Quad. Shimadzu is a corporate member of the Center for Sensory Sciences & Innovation.
Rachel Lee conducted the study as a George H. Cook undergraduate research scholar. We thank the panelists for their time and commitment and Tejasvi Terpuseema for her work on the early development of the lexicon. This research was approved by the Rutgers University Arts and Sciences Institutional Review Board for the protection of human subjects in research. The panelists provided written informed consent for their participation in this project.